Top 10 Strong Base Anion Resins and Their Applications?
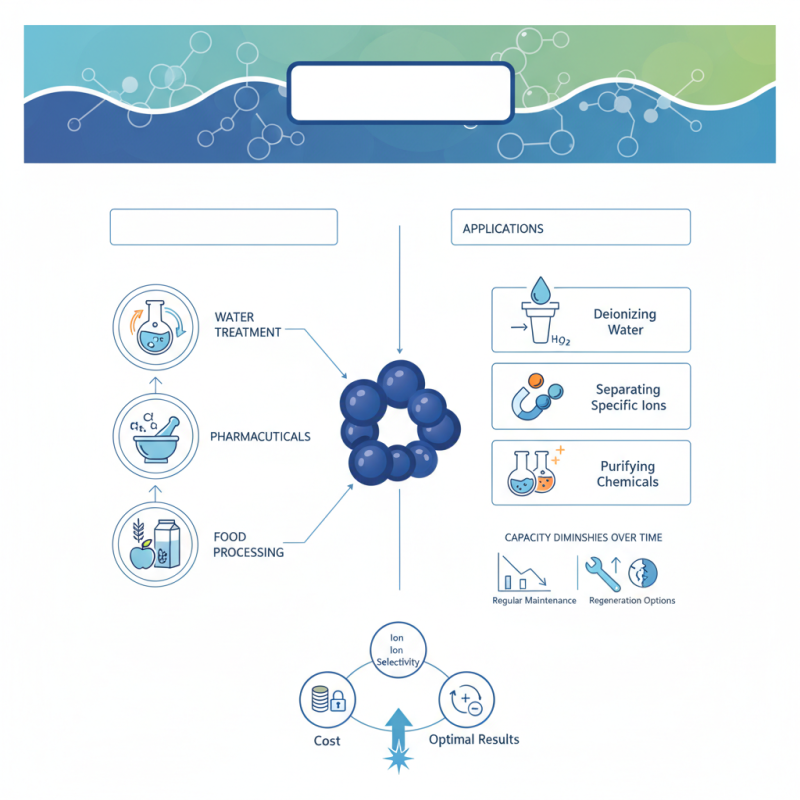
strong base anion resins play a crucial role in various industrial processes. These resins help remove unwanted ions from solutions. They are vital in water treatment, pharmaceuticals, and food processing. With their robust performance, strong base anion resins ensure high purity levels.
The applications of these resins vary significantly. They can be used to deionize water, separate specific ions, and purify chemicals. Despite their advantages, some limitations exist. For instance, the resin's capacity may diminish over time. Regular maintenance is key to ensure efficiency and longevity.
Understanding the intricacies of strong base anion resins is essential. Each type has its unique characteristics and uses. Choosing the right resin can be challenging. Decision-makers must consider various factors, such as cost, ion selectivity, and regeneration options. The right choice leads to optimal results and efficiency.
Overview of Strong Base Anion Resins
Strong base anion resins are crucial in various industries. They play a vital role in water treatment, biopharmaceuticals, and chemical processing. These resins facilitate the removal of anions, leading to purified water and enhanced product quality.
In water treatment, strong base anion resins effectively remove harmful substances. They also help in the deionization process. Their efficiency is high, but their regeneration needs careful handling. This step can sometimes be overlooked, leading to resin fouling.
Tips: Always monitor the resin’s performance. Regular checks help in maintaining its efficiency. If performance declines, consider a thorough cleaning or replacement.
In biopharmaceuticals, these resins support the purification of proteins and peptides. Their selectivity makes them suitable for specific applications. However, it’s important to choose the right resin type for optimal results. The learning curve can be steep.
Tips: Keep an eye on operational conditions. Temperature and pH can affect performance. Experiment with setup adjustments to find the best configuration.
Characteristics of Top Strong Base Anion Resins
Strong base anion resins play a crucial role in various industrial applications. These resins are known for their unique ability to remove anions from solutions. They are typically made from polystyrene and are chemically designed to withstand harsh environments. The strong base functionality allows them to interact effectively with a variety of contaminants.
One key characteristic is their high exchange capacity. This feature enables the resins to process large volumes of water quickly. In water treatment, this can lead to improved purity levels. Additionally, they are resistant to degradation. This durability ensures a longer lifespan and less frequent replacements.
However, not all applications yield the same results. Some resins may not perform optimally under specific conditions. Temperature, pH levels, and specific contaminants can greatly affect their efficiency. Choosing the right resin requires careful consideration of these factors.
It’s essential to keep in mind that the performance of strong base anion resins can vary significantly depending on the environment.
Applications in Water Treatment and Purification
Anion resins play a crucial role in water treatment and purification. These materials are essential for removing unwanted contaminants and ions. They effectively address issues like hard water and dissolved organic matter. According to industry reports, over 70% of water treatment facilities utilize anion exchange resins for improving water quality.
The effectiveness of anion resins is driven by their ability to exchange negatively charged ions. These resins help in removing sulfate, nitrate, and phosphates. In some cases, however, they may not provide complete removal, requiring additional treatment steps. The efficiency can vary depending on the resin type and water chemistry. This variability points to the need for ongoing optimization in treatment processes.
Recent studies indicate that using these resins can reduce contaminants by up to 90% in specific applications. Yet, challenges remain. Resin fouling and saturation can limit performance. Regular maintenance and monitoring are crucial to sustain effectiveness. The quest for finding the right balance between cost, efficiency, and eco-friendliness continues. Choosing the ideal resin for a specific application is pivotal in achieving the desired water quality.
Industrial Uses of Strong Base Anion Resins
Strong base anion resins have various industrial applications. These resins effectively exchange anions in solutions, making them essential in water treatment. They are commonly used in deionization processes to remove harmful anions. Industries rely on them to ensure water quality. They help in producing high-purity water for pharmaceutical and electronics manufacturing.
Another significant application involves wastewater treatment. Strong base anion resins assist in the removal of contaminants, including nitrates and sulfates. They play a crucial role in environmental protection. By treating industrial waste, these resins minimize harmful effects on ecosystems. This task is not straightforward, and efficiency can vary based on many factors.
Moreover, these resins are used in food and beverage production. They help in clarifying liquids and removing unwanted tastes. This ensures products meet quality standards. However, not all resins perform equally in every situation. Understanding their limitations is essential for optimal use. This ongoing learning process can enhance their effectiveness in various settings.
Top 10 Strong Base Anion Resins and Their Applications
Comparison of Performance and Cost Factors
When considering strong base anion resins, performance often shapes their application. These resins vary significantly in their ion exchange capacities. Some excel in removing specific contaminants, while others are more versatile. For instance, certain resins effectively capture heavy metals. Yet, the choice may not always be straightforward.
Cost remains a critical factor. Some resins deliver high performance but come with a hefty price tag. In contrast, other options offer acceptable results at lower costs. It's essential to weigh these aspects carefully. Relying solely on price may overlook long-term efficiency. A resin that seems cheap might require frequent replacements, leading to higher costs over time.
Furthermore, not all applications will suit the most expensive resin. Sometimes, a mid-range option meets the need perfectly. Efficiency doesn't always correspond to price. Users may face dilemmas when evaluating their options. Performance metrics can be tricky to interpret without proper context. Making the right choice may require deeper analysis. The balance between cost and performance remains a challenging but vital undertaking.
Top 10 Strong Base Anion Resins and Their Applications
| Resin Type | Ion Exchange Capacity (meq/mL) | Operational pH Range | Max Operating Temperature (°C) | Common Applications | Cost per Liter (USD) |
|---|---|---|---|---|---|
| Type I Strong Base Anion | 1.2 | 0 - 14 | 100 | Water Purification, Waste Treatment | 50 |
| Type II Strong Base Anion | 1.4 | 0 - 14 | 120 | Pharmaceuticals, Chemical Processing | 55 |
| Macroporous Anion Resin | 1.1 | 1 - 10 | 80 | Food and Beverage Treatment | 70 |
| Quaternary Ammonium Anion Resin | 1.3 | 2 - 12 | 90 | Biosciences, Chemical Analysis | 65 |
| Functionalized Anion Resin | 1.5 | 0 - 14 | 100 | Industrial Wastewater Treatment | 75 |
| Cation-Exchange Modified Anion Resin | 1.0 | 4 - 10 | 70 | Mining Effluent Treatment | 60 |
| Chloride Anion Exchange Resin | 1.2 | 0 - 14 | 95 | Chemical Purification, General Water Softening | 50 |
| Ethanol-resistant Anion Resin | 1.5 | 4 - 11 | 85 | Beverage Industry, Ethanol Cover-strain | 80 |
| Hybrid Anion Resin | 1.6 | 0 - 14 | 120 | Power Plant Water Treatment | 100 |
| Superior Performance Anion Resin | 1.7 | 0 - 14 | 150 | Advanced Chemical Processing | 120 |
